Pioneering solutions, saving lives
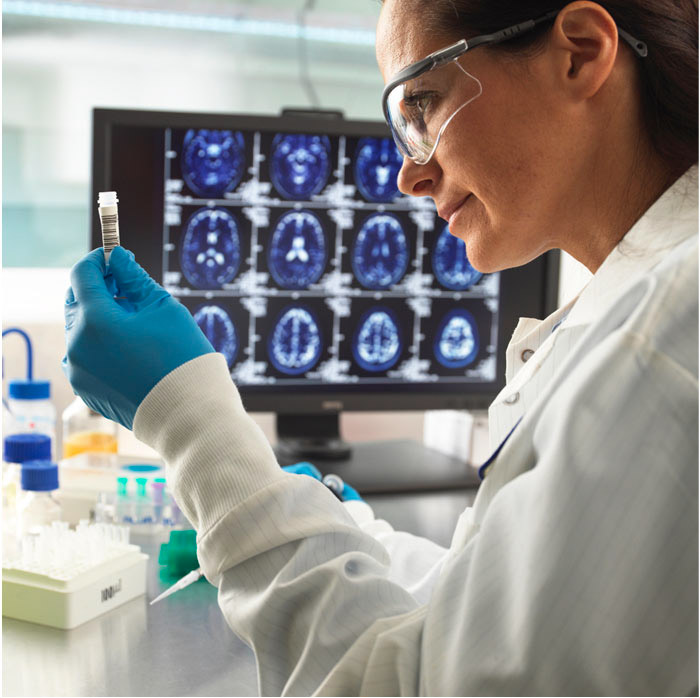
Research is critical to combatting brain disease and improving the lives of those impacted, today and in the future.
Conducted by The Royal Melbourne Hospital’s Departments of Neurology and Neurosurgery, the active research programs and clinical trials supported by the Foundation cover an extensive breadth of neurological conditions.
Patients wishing to participate are encouraged to view the relevant trial and contact the trial co-ordinator for more information.
| Brain Tumour / Neurosurgery: | NsurgClerk@mh.org.au |
| Dementia: | DementiaTrials@mh.org.au |
| Epilepsy: | EpilepsyCoordinator@mh.org.au |
| Huntington’s Disease: | RMHHuntingtonDiseaseStudies@mh.org.au |
| Movement Disorders: | PDNurse@mh.org.au |
| Multiple Sclerosis: | MSClinic@mh.org.au |
| Neurophysiology: | RMH-ClinicalNeurophysiology@mh.org.au |
| Stroke: | RMH-StrokeResearchTeam@mh.org.au |
| General: | Neuro.Foundation@mh.org.au |
A Phase 3, Double-blind, Randomized, Placebo-controlled, Parallelgroup, Multicenter Study to Evaluate the Safety, Tolerability, and Efficacy of 2000 mg/kg of Trappsol® Cyclo™ (Hydroxypropyl-β-cyclodextrin) and Standard of Care Compared to Placebo and Standard of Care in Patients with Niemann-Pick Disease Type C1

Prospective Neuro-Ophthalmology database (NODE) towards developing comprehensive longitudinal outcome measures for neuro-ophthalmic diseases
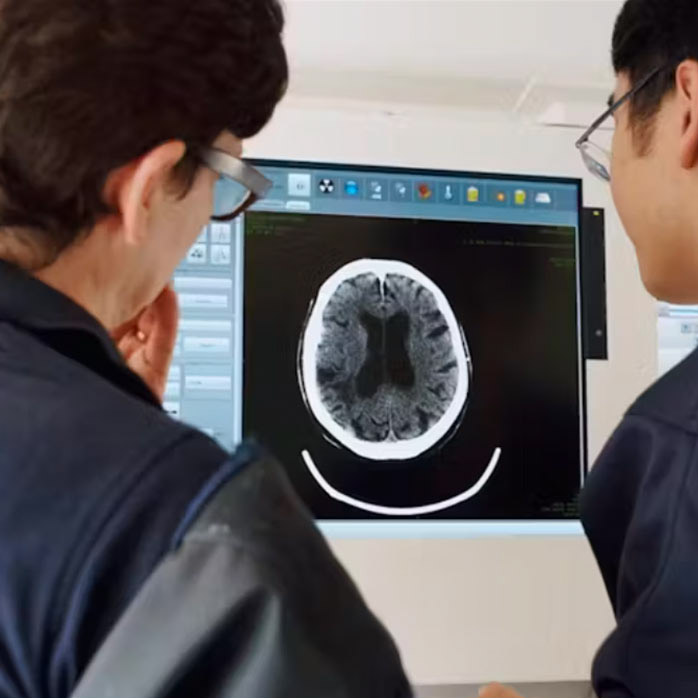
A Phase II, Multicenter, Double Blinded, Randomized, Placebo Controlled, Parallel-Group, Single-Dose Study to Determine the Safety, Preliminary Efficacy,and Pharmacokinetics of ARG 007 in Acute Ischemic Stroke Patients

Safety and Tolerability of Adjunctive TBO-309 in Reperfusion for Stroke

"A multicenter, international, randomized, placebo controlled, double-blind, parallel group and event driven Phase 3 study of the oral FXIa inhibitor asundexian (BAY 2433334) for the prevention of ischemic stroke in male and female participants aged 18 years and older after an acute non-cardioembolic ischemic
stroke or high-risk TIA"

