Pioneering solutions, saving lives
Research is critical to combatting brain disease and improving the lives of those impacted, today and in the future.
Conducted by The Royal Melbourne Hospital’s Departments of Neurology and Neurosurgery, the active research programs and clinical trials supported by the Foundation cover an extensive breadth of neurological conditions.
Patients wishing to participate are encouraged to view the relevant trial and contact the trial co-ordinator for more information.
| Brain Tumour / Neurosurgery: | NsurgClerk@mh.org.au |
| Dementia: | DementiaTrials@mh.org.au |
| Epilepsy: | EpilepsyCoordinator@mh.org.au |
| Huntington’s Disease: | RMHHuntingtonDiseaseStudies@mh.org.au |
| Movement Disorders: | PDNurse@mh.org.au |
| Multiple Sclerosis: | MSClinic@mh.org.au |
| Neurophysiology: | RMH-ClinicalNeurophysiology@mh.org.au |
| Stroke: | RMH-StrokeResearchTeam@mh.org.au |
| General: | Neuro.Foundation@mh.org.au |
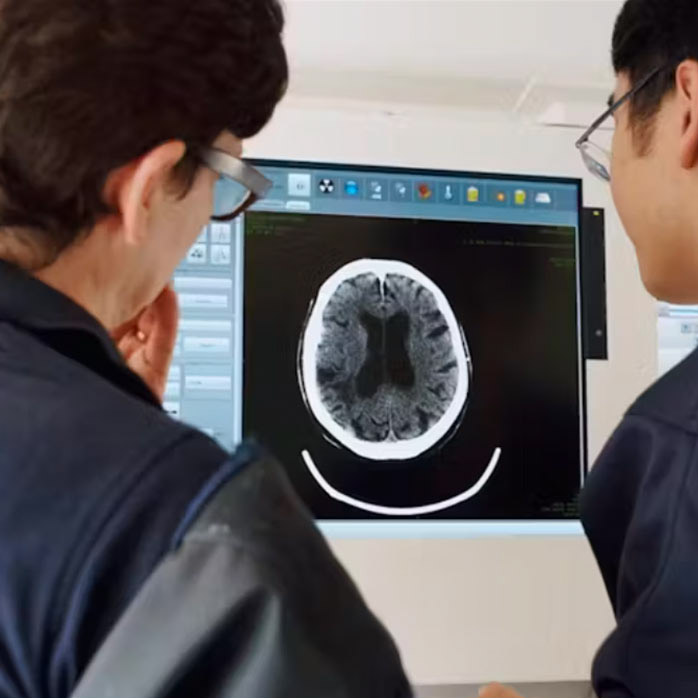
LOw dose combinations To improve stroke oUtcomeS (LOTUS)
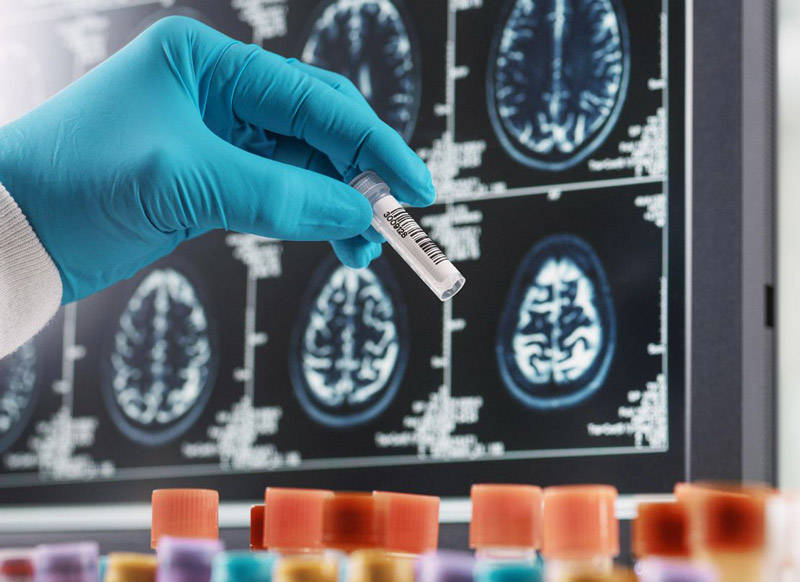
CLOU064C12306: A randomized, open-label, parallel-group, non-inferiority study comparing efficacy, safety, and tolerability of remibrutinib after switching from ocrelizumab in participants living with relapsing multiple sclerosis, followed by open-label treatment with remibrutinib

A multi-faCtorial, mulTi-arm, multi-staGe, randomised, gLOBal pLatform trial for Stroke (ACT-GLOBAL)
An Open-label Study to Assess the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of GT-02287 in Participants with Parkinson’s Disease With or Without a Pathogenic GBA1 Mutation
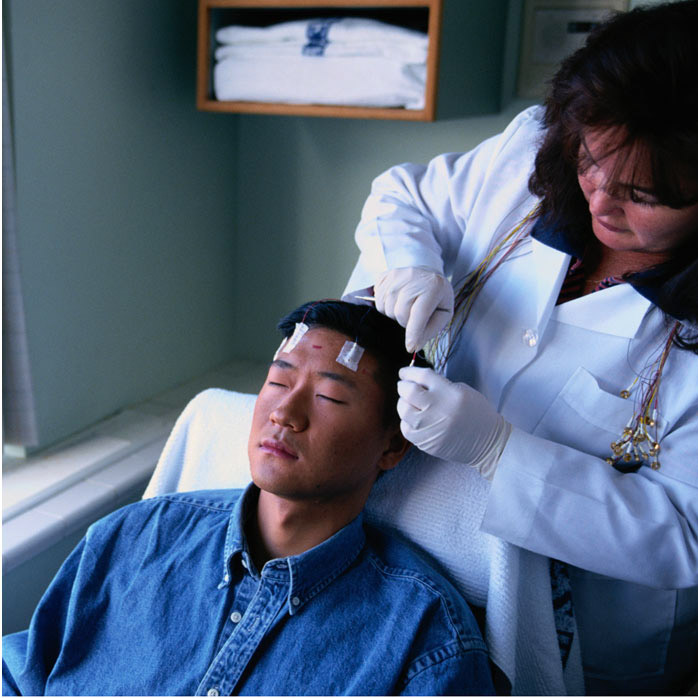
A Randomized, Double-blind, Placebo-Controlled, Multicenter Phase 3 Study to Evaluate the Safety, Tolerability, and Efficacy of XEN1101 as Adjunctive Therapy in Focal-Onset Seizures (XTOLE3)

